Researchers Break Decades-old Bottleneck in Chemotherapy Drug Manufacturing
New method produces 180% more doxorubicin than current industrial standards
We have harnessed rational strain engineering to pave the way for cost-effective manufacturing that can meet growing global demand.”
TURKU, FINLAND, March 13, 2026 /EINPresswire.com/ -- An international team of researchers has achieved a breakthrough in the production of doxorubicin, a vital chemotherapy agent. The study identifies and resolves molecular "bottlenecks" that have limited the natural production of this drug for over 50 years.— Dr Keith Yamada
Doxorubicin is a chemotherapy drug that was first approved for medical use in the 1970s. It is a cornerstone in treating various cancers, including breast cancer, bladder cancer, lymphomas and carcinomas, with over one million patients receiving the treatment annually. However, bacteria naturally produce this important drug very inefficiently. Consequently, the pharmaceutical industry has relied on expensive, multi-step semi-synthetic processes.
"We have uncovered several independent factors that limit the formation of doxorubicin," says Researcher Keith Yamada, PhD, from the University of Turku in Finland, a lead scientist on the study. "By addressing these bottlenecks, we have harnessed rational strain engineering to pave the way for cost-effective manufacturing that can meet growing global demand".
Researchers Develop New Strain of Bacteria that Enhances Drug Production
The study was the result of an extensive international collaboration involving a total of six research laboratories: the University of Turku in Finland, three laboratories in the United States, and two in Leiden, the Netherlands.
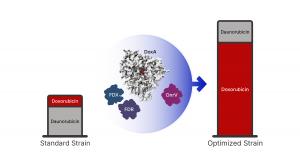
Together, the teams identified three primary constraints that prevent the high-yield production of doxorubicin.
Firstly, the team identified the specific natural "biological power supply" — redox partners named Fdx4 and FdR3 — that provide the necessary electron flow to power the drug-producing enzyme.
Secondly, they discovered that a protein called DnrV acts as a drug-binding "molecular sponge". It sequesters (binds and holds) doxorubicin so the drug does not shut down the enzyme's own production machinery.
Last, using X-ray crystallography, the team visualized the enzyme for the first time, revealing that the drug molecule sits in an unfavorable position within the enzyme, explaining the slow reaction rate.
By combining these discoveries, the researchers engineered a new strain of bacteria that produces 180% more doxorubicin than current industrial standards.
To bring these findings to the real world, the spin-out company Meta-Cells Oy was formed last year at the University of Turku. The company aims to commercialize these advanced technologies for the sustainable manufacturing of essential antibiotics and anti-cancer agents. This shift toward fully biosynthetic production promises a cleaner, more reliable supply of life-saving medicines.
The study was published in the renowned journal Nature Communications.
Keith Yamada
University of Turku
communications@utu.fi
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.